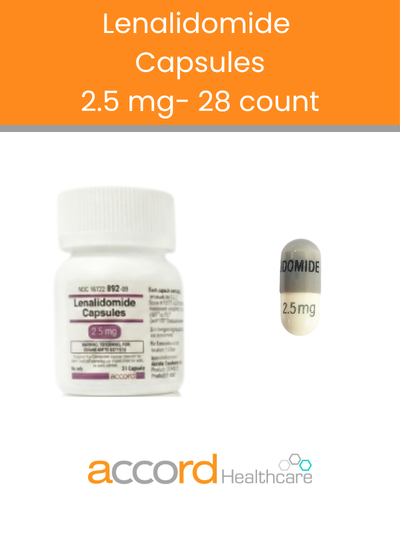
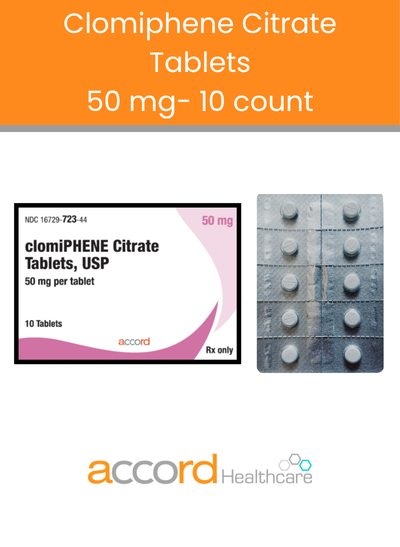
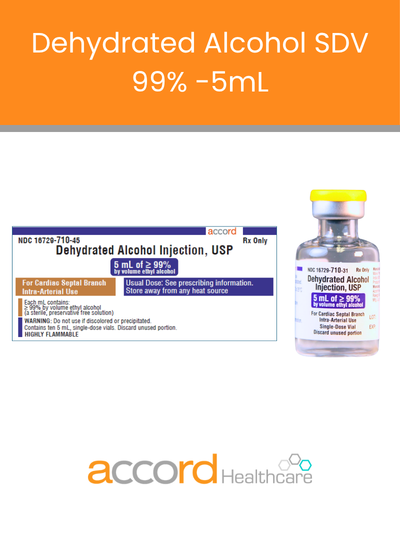
Welcome to Accord Healthcare
We are reimagining the generic pharmaceutical industry so that value, service, safety, and innovation are integrated to create well-being for all.
Accord Healthcare, Inc., the US subsidiary of Intas Pharmaceuticals, is a leading generic pharmaceutical company headquartered in Durham, North Carolina. Through its subsidiaries, Intas markets its products in 80 countries.
We understand the importance of empowering physicians and patients by providing access to the highest quality products. Our passion and commitment to improving lives by delivering timely and affordable solutions that meet important medical needs has earned us a reputation for outstanding quality, value and service.
Get to know us!
Research And Development
Committed. Integrated. Global.
Our scientists, researchers, and healthcare professionals, apply their expertise through our state-of-the-art labs and manufacturing infrastructure. Through their expertise, we can successfully navigate regulatory pathways and advance the right environment for true innovation. Accord Healthcare is among the top 10 generic players by prescription.*
Our pipeline spans a full range of dosage forms, including orals, topicals, liquid, lyophilized and complex injectables.
As a leader in oncology, Accord Healthcare constitutes a 30% volume share of major chemotherapy injectable molecules. Across all areas, our excellence in research is driven by 500 scientists involved in product development across the globe.
*IQVIA NSP MAT April 2022Manufacturing
Accord Healthcare has three FDA-approved facilities based in India. Matoda and SEZ are both located in our home city of Ahmedabad. These facilities have the flexibility to manufacture various dosage forms in a wide range of batch sizes.
As we continue to expand and grow our portfolio, Accord Healthcare is meeting and surpassing expectations in our market. With an excellent regulatory track record, we have successfully completed 130 inspections in the last 10 years across our manufacturing facilities, including 22 inspections by the US FDA.